Trust your critical cells to a company with over 15 years iPSC differentiation experience
Combining the convenience of cell-based models with the biorelevance of primary cells, iPSC-derived cell lines are a scalable resource for disease modeling, drug discovery and screening, and regenerative medicine.
With our comprehensive iPSC differentiation services, you can confidently leverage the power of iPSC-derived cells for your own insights. We offer progenitor and lineage-committed cell types, customizable characterization, and can even integrate your differentiation service with iPSC genome engineering as well as cell banking.

iPSC-derived cortical neurons (staining for Tuj1 (red) and DAPI (blue)).
With hypoimmunogenic iPSCs and integrated iPSC genome engineering, differentiation, and cell banking services, we give you a streamlined path to IND.
We specialize in many different lineages—neural, hematopoietic, cardiovascular, photoreceptors, and more. Don’t see a lineage you are interested in—just ask.
We also offer customized differentiation protocol development for lineages not listed.
Neural & glial cells
Blood, immune, & hematopoietic cells
Hepatocytes
Cardiac & muscle cells
Retinal cells
MORE
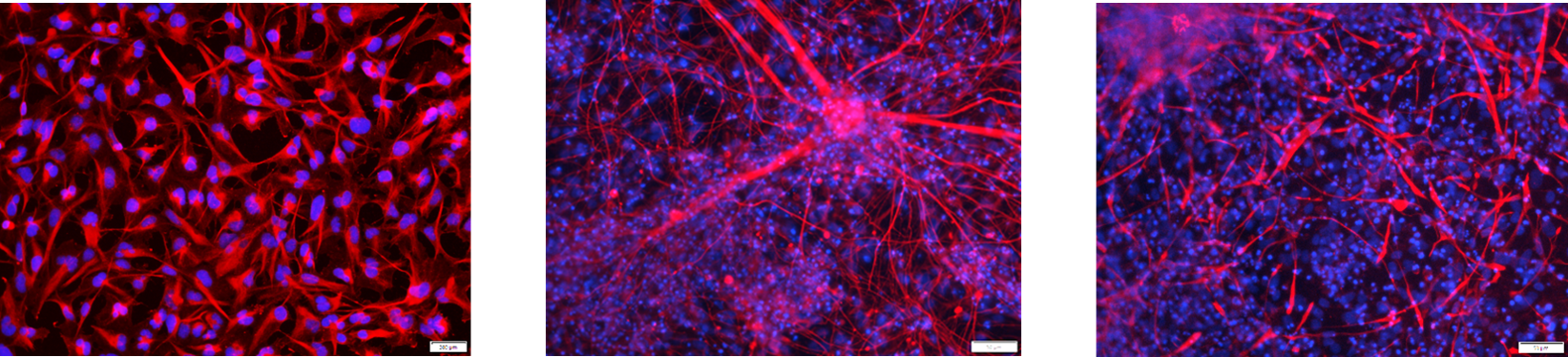
Immunostained NPCs (left panel), neurons (middle panel), and astrocytes (right panel). We differentiated iPSCs (Cat.# ASE-9211) into NPCs (nestin (red), nuclei (DAPI, blue), and then neurons (Tuj1 (red), nuclei (DAPI, blue)) and astrocytes (GFAP (red), nuclei (DAPI blue)).

Brightfield image of astrocytes we differentiated from reprogrammed fibroblasts (Cat.# ASE-9211)

The cells shown in the brightfield image exhibited strong and uniform staining for the astrocyte markers GFAP (left panels) and s100beta (right panels). Left panels: GFAP staining (top, green), nuclear staining with DAPI (middle, blue), GFAP/DAPI merge (bottom).
Right panels : s100beta staining (top, red), nuclear staining with DAPI (middle, blue), s100beta/DAPI merge (bottom).

Dopaminergic neurons derived from reprogrammed fibroblasts (Cat.# ASE-9211). Cell identity demonstrated by staining with tyrosine hydroxylase (TH, green).
Additional characterization is shown below through staining with corin, OTX2, and nestin.

Asher MJ, McMullan HM, Dong A, Li Y, Thayer SA. A Complete Endocannabinoid Signaling System Modulates Synaptic Transmission between Human Induced Pluripotent Stem Cell–Derived Neurons. Mol Pharmacol. 2023;103(2):100-112. doi:10.1124/molpharm.122.000555
Gupta G, Gliga A, Hedberg J, et al. Cobalt nanoparticles trigger ferroptosis‐like cell death (oxytosis) in neuronal cells: Potential implications for neurodegenerative disease. FASEB J. 2020;34(4):5262-5281. doi:10.1096/fj.201902191RR
Jurkovicova-Tarabova B, Stringer ,Robin N., Sevcikova Tomaskova ,Zuzana, and Weiss N. Electrophysiological characterization of sourced human iPSC-derived motor neurons. Channels. 2025;19(1):2480713. doi:10.1080/19336950.2025.2480713
Wang R, Maloney B, Beck JS, Counts SE, Lahiri DK. MicroRNA-153-3p targets repressor element 1-silencing transcription factor (REST) and neuronal differentiation: Implications for Alzheimer’s disease. Alzheimers Dement. 2025;21(8):e70399. doi:10.1002/alz.70399
Lin SY, Wu TH, Yao CN, et al. Priming cell-degradable ubiquitination of Aβ42 with molecular crosstalk. Research Square. Preprint posted online March 25, 2025. doi:10.21203/rs.3.rs-5961309/v1
Find out how our iPSC Differentiation Services can fuel your work by submitting the form below. We will get back to you within one business day.